3141 / 157 inorganic liquid compouns, n.o.s.Vapor Pressure = C Regulatory / Health.Flammablity Class: Non-combustible solid (except as dust).Enthalpy of Atomization: 263.6 kJ/mole 25☌. 
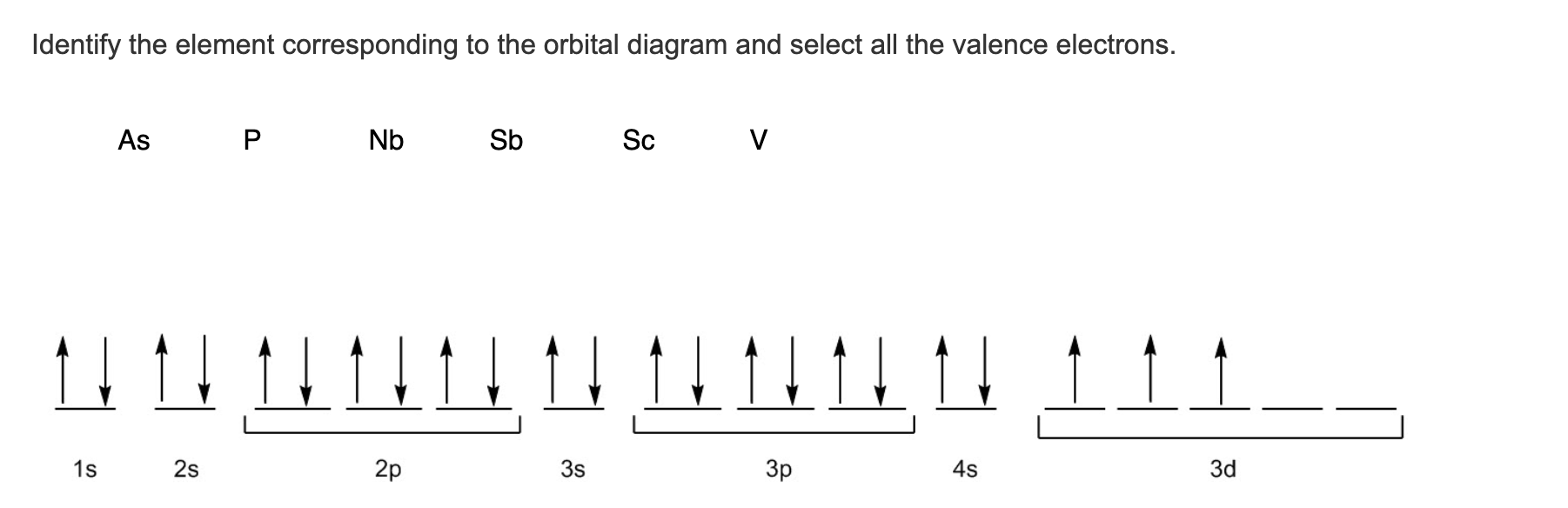
Description: Hard brittle bluish-white metal.Conductivity Electrical: 0.0288 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 8.5E -6.Valence Electron Potential (-eV): 57 Physical Properties of Antimony.Incompatibilities: Strong oxidizers, acids, halogenated acids.Electronegativity: 2.05 (Pauling) 1.82 (Allrod Rochow).Electrochemical Equivalent: 1.5142g/amp-hr.Valence Electrons: 5s 2p 3 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 71.Number of Electrons (with no charge): 51.Electrons per Energy Level: 2,8,18,18,5 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 10 4s 2p 6d 10 5s 2p 3.Cross Section (Thermal Neutron Capture) σ a/ barns : 4.91.Swedish: Antimon Atomic Structure of Antimony.Series: Metalloids ( Metals) Antimony's Name in Other Languages.Common Chemical Compounds of Antimony Overview of Antimony.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Antimony - SbĬomprehensive data on the chemical element Antimony is provided on this page including scores of properties, element names in many languages, most known nuclides of Antimony.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
